Low levels of return on investment in R&D is an issue that has been bubbling beneath the surface of pharmaceutical companies for some time and the situation is coming to a head, threatening the industry’s future
Words by James Coker
Medical intelligence is a concept increasingly being pointed to as a means of addressing this challenge, but first and foremost, it must be clearly defined. “When you think of intelligence you envision military intelligence”, outlines Isma Benattia, Vice President, Europe Medical Affairs, Amgen. “This critical discipline is constructed on information collection and analysis to guide and assist commanders in their decisions. Medical intelligence is no different. We need to apply the same discipline in data collection and interpretation of clinical, biotechnology, and environmental information to guide R&D strategies and resource planning.”
This highly methodical approach described by Benattia is no mere fad for pharma to try, nor is it the latest buzzword to be echoed around the industry’s walls; it is the bricks and mortar upon which future R&D strategies should be constructed, with MA playing the role of chief architect. “As MA is well positioned to understand, engage, and support both the patient and provider journey, it becomes all the more essential for MA to steward the trusted, valid, and reliable ‘source of truth’ and relationships associated”, notes Jennifer Wong, Senior Director, Real World Evidence Strategy & Alliances, Global Medical Affairs, AstraZeneca.
A disciplined approach to medical intelligence will allow an early detection of unmet needs
Using this intelligence-led approach is critical for enabling R&D teams to focus on areas that can have the most profound impact for patients and consequently, improve return on investment. “A disciplined approach to medical intelligence will allow an early detection of unmet needs”, explains Benattia. “The close contact with data collection and analysis of the healthcare ecosystem, which is in constant evolution, will allow the detection of early signals of changes in healthcare.”
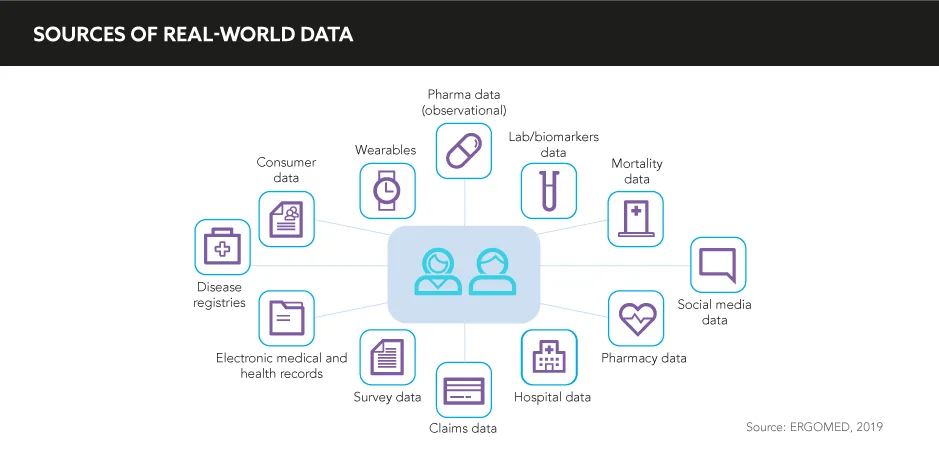
Real-world evidence, in particular, provides a seemingly bottomless pit of health information that can identify the best opportunities for pharma, and MA are now required to dig deep beneath the surface to discover as many golden nuggets of information as possible. “MA is in a unique position within the pharma industry to impact drug development from bench to bedside. When utilised properly, patient registries, claims databases, and medical records are rich sources of real-world data that can inform R&D strategies”, says Wendell Valdecantos, Director, Immunology Clinical Development and Medical Affairs, Boehringer Ingelheim.
Access to vast swathes of data is one thing; obtaining relevant insights from this data is an entirely different affair. Firstly, heavy investment in state-of-the-art digital technologies in data analytics is necessary to ensure MA can perform this function properly. “We are undergoing a technical and cultural transformation”, comments Wong. “Keeping up with the latest advanced technologies is both exciting and daunting due to the size and scale of the options available. Most importantly, we all need to be empowered and well-equipped to learn, assess, adapt, and take action quickly and intelligently and cut through the noise to identify, curate, and act on ‘actionable information’ to drive real-world decisions and outcomes.”
MA is in a unique position within the pharma industry to impact drug development from bench to bedside
Once the technology is in hand, MA must then become adept at using it, which is no mean feat. It requires new skills that go beyond their traditional requisite of medical knowledge and good communication. “We need to consider diversifying MA’s conventional talent pool”, says Benattia. “We have to augment the scientific expertise with digital and analytical ones and add these threads to the MA fabric. It encompasses digital skill and monitoring solutions for big data mining. The aim is to detect early new trends in patients’ care and turn them into insights to guide research activities. These investments in the development of the MA staff will allow the use of the new technologies to their maximum effect.”
The vast availability of data, coupled with sophisticated data-mining technologies, gives pharma a unique chance to revolve R&D strategies around unmet needs which provide the best opportunities to improve return on investment. This also offers a long-term solution to the recent decline in productivity observed throughout the industry in the last decade. MA should be at the heart of this methodical, intelligence-based approach, and be afforded the data analytic technology and technical skills required to accurately present this information to R&D colleagues.